An estimated 1 billion people between the ages of 30 and 69 across the globe suffer from sleep apnea, particularly obstructive sleep apnea, according to a 2019 study. To treat sleep apnea, healthcare providers typically suggest using a continuous positive airway pressure (CPAP) device or neuromuscular electrical stimulation (NMES), the latter of which keeps the tongue and upper airway muscles from obstructing breathing during sleep. Clinicians may also suggest lifestyle changes, the treatment of underlying conditions that exacerbate airway obstruction, or, in more severe instances, surgery.
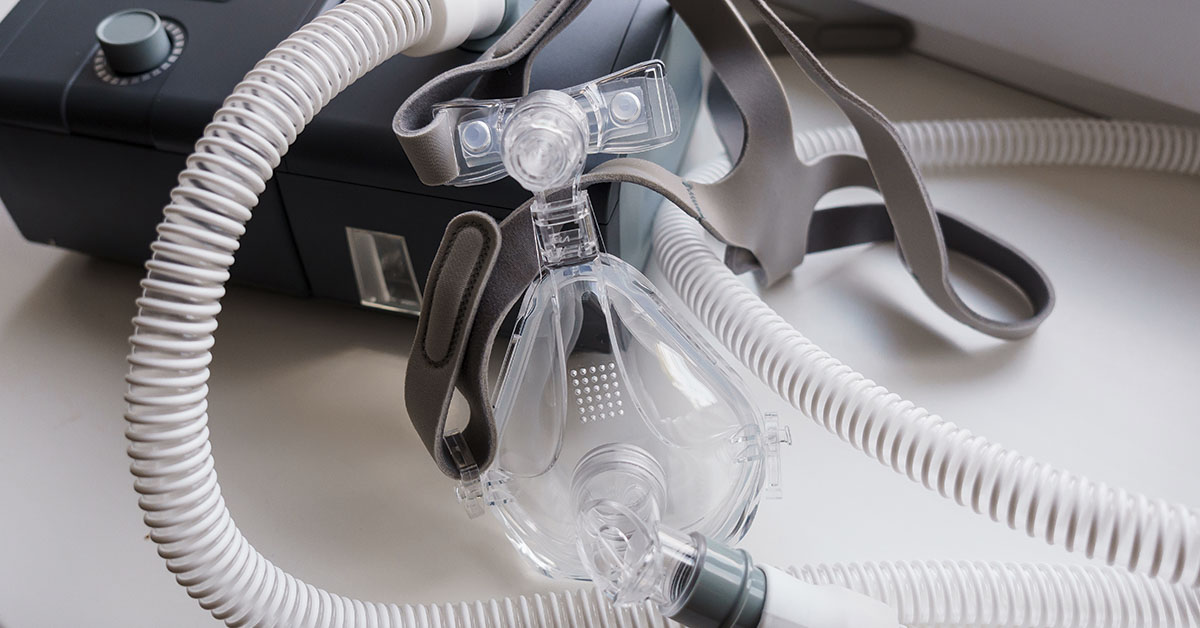
Current sleep apnea therapies effectively manage the disorder but often prove both invasive and costly. CPAP machines, which maintain open airways by delivering continuous air pressure to the lungs, serve as a primary example. These devices demand significant amounts of maintenance, and the ongoing expense of replacement components can be costly. In the near future, however, a newly developed pill could render CPAP machines and other costly, intensive sleep apnea treatments obsolete.
According to a clinical trial featured in The Lancet, a breakthrough in treating obstructive sleep apnea may be on the horizon. The study found that sulthiame, an anti-seizure medication repurposed for this new use, reduced breathing disruptions in patients by as much as 47%. This finding offers significant promise for the estimated 1 billion individuals globally affected by the condition.
What Sleep Apnea Actually Does to the Body

Obstructive sleep apnea occurs when the upper airway muscles relax while asleep. When the airway relaxes, it collapses, cutting off oxygen supply to the lungs. The brain then jolts the body awake just long enough to resume breathing before the cycle repeats itself. This pattern can repeat dozens of times per hour, and the person frequently has no memory of it.
The Cognitive Toll
Aside from fatigue, the oxygen deprivation caused by repeated apnea events has a direct effect on the brain. Each episode prevents the body from entering deep, restorative sleep, which allows the brain to consolidate memories and eliminate metabolic waste. Ohio State University researchers note that the cognitive symptoms of obstructive sleep apnea can be similar to those of early-stage dementia, such as forgetfulness, difficulty planning, and slowed processing speed. Excessive daytime sleepiness affects up to 90% of those with the condition, and it is not the kind of fatigue that a single good night’s sleep will cure.
The Workplace and Road Safety Consequences
Sleep apnea is harmful to more than just one’s health. It lowers productivity and puts others in danger. According to a systematic review and meta-analysis published in the journal Sleep, workers with obstructive sleep apnea are nearly twice as likely to have a workplace accident as workers without the condition. Drivers with sleep apnea face a two to eight-fold increased risk of a motor vehicle crash when driving for work. According to researchers, obstructive sleep apnea is the single most preventable medical cause of daytime sleepiness-related driving accidents.
The Long-Term Health Cost
Repeated oxygen deprivation and interrupted sleep put a strain on the cardiovascular system. Obstructive sleep apnea is associated with a significantly increased risk of high blood pressure, coronary artery disease, heart failure, and stroke. UT Southwestern Medical Center research published in the Journal of the American Heart Association confirmed the cardiovascular link in adults under the age of 40. Sleep apnea, left untreated, increases the risk of developing type 2 diabetes, metabolic syndrome, or depression.
Why So Many Cases Go Undetected

Despite its scale, sleep apnea remains one of the most underdiagnosed conditions in medicine. According to estimates, 80 to 90% of moderate to severe cases go undetected around the world. Many patients believe that chronic fatigue, morning headaches, irritability, and mood changes are normal side effects of stress or aging. People with sleep apnea rarely associate their symptoms with nighttime breathing interruptions. Because the condition is not visible to the patient, closing the diagnostic gap is extremely difficult without proactive screening measures.
The Cost of Getting Diagnosed
Getting a formal diagnosis can be expensive. An in-lab sleep study, known as a polysomnogram, costs between $1,000 and $10,000, with the national average at around $3,000. Home sleep tests are a more affordable option, typically costing between $150 and $1,000. Even at the lowest level, the financial barrier deters many patients from seeking a diagnosis at all. Those who do get tested must pay for treatment in addition to the diagnostic fee. The American Academy of Sleep Medicine estimates that undiagnosed sleep apnea costs the United States $149.6 billion per year in healthcare costs, workplace accidents, and lost productivity. Ignoring it is far more expensive than treating it.
How the Therapy Works
By providing a continuous flow of air through a mask that covers the nose or face, CPAP machines generate the pressure needed to prevent airway collapse during sleep. This mechanical support ensures patients can breathe without interruption throughout the night. When used according to clinical guidelines, CPAP therapy is a highly effective treatment for maintaining open airways.
The Compliance Crisis
For decades, CPAP adherence has remained consistently poor, with technological advancements failing to drive significant improvement. A meta-analysis examining twenty years of data revealed that non-adherence remains around 34 percent, with overall compliance rates fluctuating between 30 and 60 percent across various studies. Some research suggests even more severe trends, with estimates indicating that up to 83 percent of patients fail to use their devices for the clinically recommended duration each night. Low rates of sustained use are particularly prevalent among women and younger patients.
The factors driving this lack of compliance are uniform across the literature: users find the masks uncomfortable, the noise levels intrusive, and the disruption to a partner’s sleep problematic. Furthermore, the logistical difficulties of traveling with the equipment create an unsustainable burden for many patients.
The Financial Burden
A standard CPAP device costs $500 to $1,000, while more advanced BiPAP machines cost $1,700 to $3,000. These figures do not include the ongoing replacement costs. Masks, filters, tubing, humidifier chambers, and cushions all need to be replaced regularly, and blower motor repairs can cost $250 to $350 alone. Patients without comprehensive insurance bear these costs directly, and the burden increases over time. Financial pressure exacerbates comfort issues and results in attrition rates that researchers have documented for over two decades.
Sulthiame: An Old Drug with a New Purpose
Clinicians began administering sulthiame in the 1960s to treat epilepsy, and it is still administered in some German-speaking countries. This long clinical history provides researchers with a comprehensive safety reference point to work from. Researchers accelerated the evaluation of sulthiame’s potential for sleep apnea treatment because it had already received approval for another condition in several countries. Repurposing an existing drug offers a practical advantage: they have already documented the early-stage safety work.
How It Stabilizes Breathing
Sulthiame belongs to a class of drugs called carbonic anhydrase inhibitors. It reduces the activity of the enzyme carbonic anhydrase, which plays a central role in the body’s regulation of acid-base balance and respiratory drive. By inhibiting that enzyme, sulthiame induces a mild metabolic acidosis, which in turn increases the brain’s drive to breathe. A stronger and more consistent respiratory drive means the brain sends steadier signals to the muscles of the upper airway, reducing the likelihood that those muscles will relax and collapse during sleep. The drug addresses the neurological root cause of the problem rather than bypassing it with mechanical pressure, which is the critical distinction from CPAP.
A Measurable Effect on Biomarkers
Researchers looked at how the drug changed biological markers of oxygen-related stress as well as changes in breathing patterns. Hypoxia-inducible factor-1α levels go down, according to a study that came out in BMC Pulmonary Medicine in late 2024. Usually, these protein levels rise when the body goes through repeated periods of not getting enough oxygen. Those decreases stayed in place for 2 weeks after the patients stopped taking the drug. This suggests that treatment may have long-lasting effects on the body even after it is stopped. The improvements in breathing frequency and oxygen levels happen at the same time, giving researchers 2 separate signals that point in the same direction.
What the Clinical Trial Found
The study enrolled 298 adults with untreated moderate to severe obstructive sleep apnea from 28 clinical sites in 5 European countries. Researchers used a randomized, double-blind, placebo-controlled design, which meant that neither participants nor researchers knew who received the active drug at any time during the trial. Researchers administered 1 of 3 doses (100 mg, 200 mg, or 300 mg) or a placebo to participants once a day at bedtime for 15 weeks. This design methodology minimizes the risk of results being distorted by expectation or reporting bias.
A Dose-Dependent Response
The trial’s primary outcome was the relative change in the apnea-hypopnea index, a standard measure of breathing interruptions per hour, from baseline to week 15. Sulthiame met the primary endpoint with all 3 doses. Patients on higher doses had up to 47% fewer breathing interruptions while sleeping compared to those on the placebo. The trial also showed significant improvements in oxygen desaturation index scores and mean overnight oxygen saturation levels. Patients receiving higher doses reported better sleep quality and less daytime fatigue. A clear dose-response relationship emerged, with higher doses producing proportionally stronger results in every measured metric.
The Safety Picture
The FLOW trial yielded largely encouraging results regarding the drug’s tolerability, with the majority of reported side effects being both transient and minor. While prior investigations into a 400 mg dosage had noted frequent occurrences of intermittent paresthesia, a “pins-and-needles” sensation, which prompted some participants to drop out, the FLOW study used lower concentrations of 100 mg, 200 mg, and 300 mg. These reduced doses demonstrated a significantly improved safety profile. Jan Hedner, a lead researcher and senior professor of pulmonary medicine at the University of Gothenburg’s Sahlgrenska Academy, characterized these findings as a major breakthrough. Hedner and his colleagues emphasized the need for longer-term, more comprehensive clinical trials to confirm the medication’s ongoing safety and efficacy across a wider range of populations before they can submit it for regulatory approval.
The Broader Race to Replace the Mask
Other therapeutic options, aside from sulthiame, are being investigated. Apnimed, which is developing sulthiame for sleep apnea in collaboration with Shionogi, is also developing AD109, a separate compound. AD109 uses a different mechanism: instead of inhibiting carbonic anhydrase to target respiratory drive, it focuses on improving upper airway muscle tone while sleeping.
Apnimed reported successful topline results from the LunAIRo study, AD109’s second Phase 3 trial, in July 2025. At 26 weeks, the study’s primary endpoint was achieved, with a mean AHI reduction of 46.8 percent from baseline. Following these findings, the company planned to submit a New Drug Application to the FDA in early 2026, with a possible regulatory decision as early as the first half of 2027.
What That Means for Sulthiame
The progression of AD109 through Phase 3 provides relevant context for sulthiame. The same joint venture is developing both drugs, and their distinct mechanisms of action enable future combination therapy or patient-matched prescribing. According to Apnimed CEO Larry Miller, sulthiame’s differentiated mechanism has the potential to broaden the reach of oral treatment beyond the patient populations that AD109 alone can address. The two drugs may serve overlapping but distinct patient populations, depending on the anatomy and physiology that causes each patient’s sleep apnea.
What Comes Next
A Phase 2 trial confirms a drug’s potential and helps to establish safe dosing ranges. It does not guarantee long-term safety or clinical suitability across a wide range of patient populations. Researchers at the University of Gothenburg and Apnimed agree that larger Phase 3 trials are required before sulthiame can be distributed to patients on a large scale. Those trials will need to determine whether the 47% reduction in breathing interruptions lasts longer than 15 weeks. They will also have to determine whether the drug works consistently across patients with varying degrees of sleep apnea, body types, and underlying causes of airway collapse.
Read More: Can ‘Dark Showering’ Actually Improve Sleep? The Viral Trend Explained
The First Drug of Its Kind
If sulthiame passes Phase 3 trials and receives regulatory approval, it will be the first drug to treat obstructive sleep apnea’s underlying cause using a pharmacological mechanism. Currently, no approved medication addresses the condition’s neurological roots. This distinction has direct consequences for the millions of patients who find CPAP unsuitable due to cost, physical discomfort, or the ongoing logistical demands of long-term device therapy. A once-daily pill taken before bed requires a fundamentally different type of treatment commitment compared to a machine that patients must wear every night, service on a schedule, clean regularly, and fund indefinitely.
A Scale Problem That Medicine Has Not Yet Solved
When the size of the population affected by this research is considered, the stakes become clearer. Sleep apnea affects approximately 936 million adults aged 30 to 69, with estimates putting the number of moderate to severe cases at 425 million globally. Up to 80% of these cases go undiagnosed. Even among patients who receive a diagnosis and begin CPAP therapy, adherence consistently falls short of the minimal threshold required for clinical benefit. Despite four decades of incremental device improvement, the existing treatment infrastructure has not solved the problem. Sulthiame provides a different type of answer, and the clinical data thus far suggests that it may be worth pursuing.
Disclaimer: This information is not intended to be a substitute for professional medical advice, diagnosis or treatment and is for information only. Always seek the advice of your physician or another qualified health provider with any questions about your medical condition and/or current medication. Do not disregard professional medical advice or delay seeking advice or treatment because of something you have read here.