Every year in the United States, approximately two thousand people receive a lung transplant. For individuals with end-stage lung disease, it is sometimes the only chance for survival. Unfortunately, the survival rate for lung transplant patients is lower than that of other organ transplants, with a five-year survival rate of about fifty to sixty percent [1].
The biggest limiting factor in successful lung transplantation is having enough suitable donors. Not only are there not enough donors in general, but many donor lungs have suffered significant damage and are therefore unusable [2].
Restoring a damaged lung to proper function can be very difficult, and sometimes impossible, but a team of researchers has developed a new experimental technique for repairing lungs using a surprising helper: a pig.
Read: Man Who Had Transplant Finds Out Later That His DNA Changed To His Donor’s
Repairing Lungs Using Pigs
Surgeon Ahmed Hozain and biomedical engineer John O’Neill of Columbia University, the lead researchers on the team, have managed to restore a damaged lung to function by sharing its circulatory system with that of a living pig [2].
This experimental procedure is based on an existing donor lung restoration technique known as ex vivo lung perfusion (EVLP), and is the result of three years of preliminary experiments. The timeline for the experiments was as follows:
2017: O’Neill and his team developed the xenogenic (cross-species) circulation platform using a pig model to extend the amount of time a lung could be held outside the body and helped damaged lungs recover [3].
2019: Biomedical engineer Gordana Vunjak-Novakovic of Columbia University and surgeon Matthew Bacchetta of the Vanderbilt Lung Institute managed to restore a damaged pig lung by attaching them to other pigs [4].
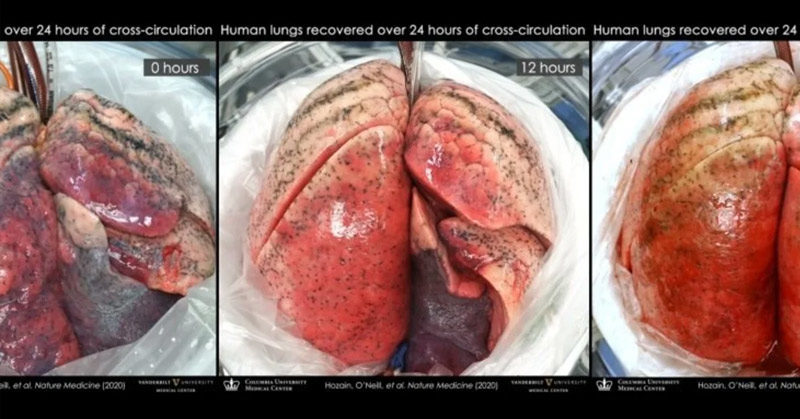
2020: The researchers have used the technique to successfully repair five damaged human lungs by connecting them to pigs [5].
Read: Bacteria that eat metal accidentally discovered by scientists
How Does the Procedure Work?
Under the current standard of care, injured lungs are restored using EVLP. With this technique, the lungs are placed in a sterile dome that is attached to a ventilator, a pump, and filters. The lung is held at human body temperature, and a solution containing oxygen, nutrients, and protein is circulated through it (a process called perfusion).
While this technique does work, it can only be conducted for up to eight hours, which is not always enough time for the biological repair functions to begin working. The pigs buy researchers more time.
For the study, the researchers obtained six donor lungs that had been rejected for transplantation. Five of the lungs were anaesthetised pigs via a jugular cannula. The pigs in the study were immunosuppressed to prevent their immune systems from attacking the lungs.
The sixth lung was attached to a pig that was not immunosuppressed, and was used as the control lung.
All of the lungs then underwent 24 hours of xenogeneic cross-circulation. The control lung broke down very quickly, as the pig’s immune system attacked it, but the other five lungs showed significant improvement in cellular viability, tissue quality, inflammatory response, and respiratory function.
One of the lungs, which had spent 22.5 hours on ice and received five hours of EVLP but showed no significant improvement, showed significantly more improvement than the researchers thought possible. The researchers said that this demonstrates that their cross-circulation platform could be used along with EVLP to save lungs that EVLP alone cannot repair.
“We were able to recover a donor lung that failed to recover on the clinical ex vivo lung perfusion system, which is the current standard of care,” Vunjak-Novakovic said. “This was the most rigorous validation of our cross-circulation platform to date, showing great promise for its clinical utility.” [2]
Read: Study Finds Vitamin D Is Linked To Low Virus Death Rate
Constraints
While this research is very promising, it is not ready to be used in a clinical setting right away. One problem with the technique is that by attaching the pigs circulatory system to a human lung, the pigs could share certain diseases with humans.
If this technique was to be used in a clinical setting, it would require medical-grade animals, but this would be much more expensive [2].
Future Possibilities
“Modifications to the xenogeneic cross-circulation circuit could enable investigation and recovery of other human organs, including livers, hearts, kidneys and limbs,” the researchers said [2].
They believe that this research opens the door to many different possibilities, including the human recipients themselves becoming the basis for the cross-circulation platform. In this scenario, the recipient would be attached to the lungs they themselves will be receiving.
The researchers see their technique being used as a translational research platform to increase transplantation research, and most importantly as a way to address the current organ shortage by salvaging previously unusable organs [2].
Keep Reading: Baby Had Experimental Surgery to Fix Spina Bifida While Still in the Womb and was Born Healthy
- https://www.uofmhealth.org/conditions-treatments/transplant/adult-lung-transplant
- https://www.sciencealert.com/a-damaged-human-lung-has-been-repaired-by-attaching-it-to-a-pig
- https://www.nature.com/articles/s41551-017-0037
- https://www.nature.com/articles/s41467-019-09908-1
- https://www.eurekalert.org/pub_releases/2020-07/cuso-sdh070820.php