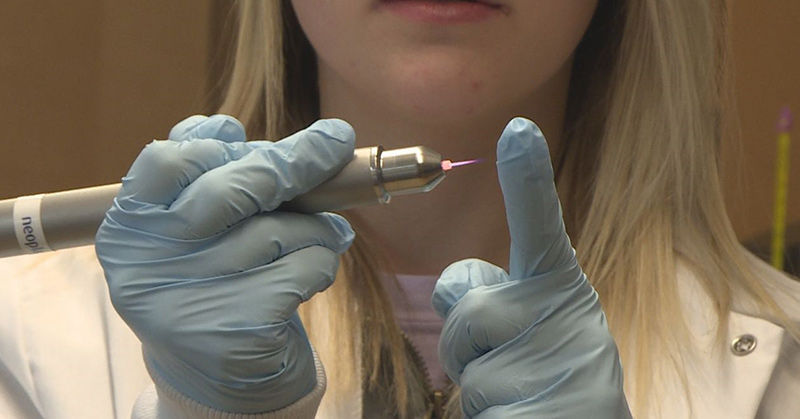Undergraduate students at the South Dakota School of Mines & Technology have been able to kill cancer cells with cold plasma, also called cold atmospheric plasma (CAP) [1].
This budding new cancer fighter wouldn’t involve drugs and has the potential to help eliminate many of the side effects that come with chemotherapy and radiation, two of the most common forms of cancer treatment today.
Overseeing the research are Prasoon Diwakar, Ph.D. and Timothy Brenza, Ph.D., of the mechanical engineering and chemical and bioengineering department, respectively. Mechanical engineering students Kristen Haller and Nicole Miller are conducting the research with the assistance of Jordan Hoops, a biological engineering Ph.D. student, and Taylor Bright, a biological sciences undergrad.
While using cold plasma to kill cancer cells isn’t a new idea (as we’ll soon see, another patented technology using cold plasma to treat cancer just went to human trials), this research is focusing on the right amount of cold plasma needed to terminate cancer cells while at the same time leaving healthy cells unaffected [2]. Here’s what we know about this developing therapy.
A Little History
Before Diwakar and Brenza began working together in 2018, Diwakar was researching cold plasma for cancer therapy at Purdue University in Indiana. Along with a professor of nuclear engineering Ahmed Hassanein, Diwakar developed Plasma Technologies or PLASMAT.
PLASMAT is a technique that uses cold atmospheric plasma with electroporation or photoporation—which we’ll talk about in a moment—to kill cancer cells but leave healthy cells intact and thriving.
Along with Brenza at South Dakota School of Mines & Technology, Diwakar worked to refine the capability of CAP to terminate just cancer cells. Their work could lead to a minimally invasive cancer treatment that would positively influence millions of patients worldwide.
Diwakar says researchers have already found a way to annihilate cancer cells using CAP, but research needs to focus more on finding the right level of cold plasma to kill cells without affecting healthy tissues. He says that finding this limit can help physicians deliver the proper dosage based on the patient, their specific type and strain of cancer, and other factors.
What Exactly Is Cold Plasma?
I don’t know about you, but when I think of cold plasma, I think of cold blood on a lab slide.
But that’s not what cold plasma is. In fact, cold plasma is quite different from blood plasma—it’s actually an ionized gas, meaning the outer electrons are separated from the atom. Cold plasma is partially ionized and has much higher energy, or charge, than normal plasma [1].
And—surprise—cold plasma isn’t actually cold, as Diwakar says. It’s actually mostly room temperature.
It’s not exactly known how cold plasma kills cancer cells, but the reaction it creates in the cells is suspected to cause death, namely between oxygen and nitrogen. However, in order to have the desired effect, the cell membrane’s pores—of which each cell has thousands—need to be opened so that cold plasma can be introduced into the inside of the cell [3].
In order to open the pores of the cell, one can use a process called electroporation, which involves putting cells in a conductive solution and running an electrical current through them for just a few milliseconds. This process alters the outer membrane of the cell, which allows its pores to open and cold plasma to be introduced into the interior.
The other process we mentioned that can be used is photoporation, which uses a laser to open pores in the cell membrane [4].
Future Potential of Cold Plasma Treatment for Cancer Cells
The students at South Dakota School of Mines & Technology hope that cold plasma could eventually replace chemotherapy to eradicate cancer cells in people. It wouldn’t have the same effect as chemotherapy or radiation on non-cancerous tissue, which means patients could avoid symptoms such as nausea, loss of hair, skin changes, and other distressing side effects [5].
Cold plasma and electroporation only currently make sense for cancers that can be easily accessed—think skin cancers or localized tumors. However, tests show that cold plasma is effective on lung cancer cells, and other research has shown potential for CAP at killing colorectal and skin cancer cells [6].
However, Diwakar says that once the treatment is proven to work, researchers will have to change how the therapy is applied. However, he says ideas are in the works, including being able to access the cancer laparoscopically, which uses smaller, minimally-invasive incisions and a camera to access surgical sites [7].
The next step, according to the researchers, is understanding what exactly it is about the reaction cold plasma creates that kills these cancer cells.
However, cold plasma is already in the works to become a potential form of cancer treatment. Human trials by the Food and Drug Administration (FDA) have already been approved and are currently underway for a prototype called the Canady Helios Cold Plasma System and Scalpel, which is a pen-shaped device that sprays cold plasma on cancer cells [2].
However, the aim for this product would not be to replace chemotherapy but to use cold plasma as an added layer to existing forms of cancer treatment such as surgery, chemotherapy, and radiation to completely destroy cancer cells—think for patients whose cancer returns, or was never fully removed in the first place. It’s never guaranteed that microscopic cancer cells—those that physicians can’t see—aren’t left behind when a tumor or other cancerous tissue is removed or destroyed [8].
What’s Next?
Clinical trials will take some time, and scientists still need to understand what it is about cold plasma that’s making these cancer cells die. In the meantime, CAP looks to be a potentially promising form of treatment for people who have cancer. As technology develops, it could treat even more difficult to reach cancers and provide hope for millions who depend on chemotherapy or radiation.
- https://www.sdsmt.edu/Research/Research@Mines/Zapping-Cancer-Cells-With-Cold-Plasma/#.XcsEHTJKj6Y
- https://www.purdue.edu/newsroom/releases/2019/Q3/treat-cancer-with-cold-plasma-purdue-aerospace-engineer-helps-bring-first-clinical-trial.html
- https://micro.magnet.fsu.edu/cells/nucleus/nuclearpores.html
- https://www.tandfonline.com/doi/full/10.1080/23746149.2016.1228476
- https://www.livescience.com/35522-side-effects-cancer-treatments-coping-tips-110302.html
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4016545/
- https://www.fascrs.org/patients/disease-condition/laparoscopic-surgery-what-it
- https://www.usmedinnovations.com/canady-life-sciences/cold-atmospheric-plasma/
- https://www.sdsmt.edu/Research/Research@Mines/Zapping-Cancer-Cells-With-Cold-Plasma/#.XcsEHTJKj6Y